Norman Chow, President
Kemetco Research, Inc.
The technology for lithium ion (Li-Ion) batteries has been available since the 1970s but there hasn’t really been a true manifestation in the marketplace until about the last 10 years. Battery makers have struggled with maintaining a significant charge, ensuring consumer safety and reducing production costs to commercially acceptable levels. But all that is about to change.
The market for Li-Ion batteries is going to explode over the next five to 10 years. Research from McKinsey Quarterly suggests that the price of Li-Ion batteries could plunge by 2020, to a more consumer friendly price point of $200 kWh (from the current $500 to $600 pricing) creating a space for the electric vehicle (including hybrid, plug-in hybrid and pure electric vehicle) market to flourish. This type of growth will first be seen in China, where Lux Research expects Li-Ion battery market will nearly double to $9.2 billion in 2016. This significant growth in the marketplace is going to place pressure on Li-Ion battery producers to create a product that is less expensive, higher performance and increasingly safer for the consumer. All three of these challenges can be met with research into a cleaner, more efficient manganese dioxide product.
In partnership with American Manganese, Inc., Kemetco Research, Inc. is working to produce improved manganese oxide material for use in rechargeable Li-Ion batteries. The research is supported by the National Research Council of Canada Industrial Research Assistance Program (NRC-IRAP) with completion of the project expected in 2013.
The Need for a Cleaner Li-Ion Material

Li-Ion batteries with cathode materials that contain manganese will have the opportunity to drive growth in the electric vehicle market. These batteries have an excellent combination of low raw materials cost, good cycle life and high specific power, which are of key importance for consideration in electric vehicles. Lithium manganese oxide batteries using LiMn2O4 chemistry are currently produced for commercial use in power tools and other consumer products. Step-wise improvements through the introduction of cleaner manganese materials will provide key advancement to the current Li-Ion battery technology for use in large format, high volume production suitable for electric vehicles. Improving known technology will drive nearer term growth.

Disruptive Li-Ion battery technologies under development, such as the 0.7 Li2MnO3 ∙ 0.3 Li4Mn5O12 layered-layered cathode chemistry developed by Agonne National Laboratory has the opportunity to more than double the specific energy of Li-Ion batteries.
Commercialization of this technology will be a game changer in providing significant boost in competitive economics and performance for large scale commercialization of electric vehicles in the future. Cleaner manganese will be of critical importance for future mass commercialization of these technologies.
As the largest battery component, the manganese supplied needs to be without metallic impurities, or can potentially contribute to thermal runaway (the over-heating of Li-Ion battery materials leading to possible fire or explosion). Thermal runaway has been a significant problem for the lithium ion battery market, one that hasn’t necessarily been a widespread issue, but has generated a public perception of Li-Ion batteries being potentially harmful. For example, in 2006 Sony, Dell, Apple, Toshiba and IBM recalled millions of laptops due to media concerns over thermal runaway in their battery packs. This recall led to millions of dollars in loss for the companies involved.
Thermal runaway continues to remain a significant hurdle for the industry today. This year, there have been recalls for Li-Ion batteries in Nikon DSLR cameras, Fisker Karma electric vehicles and HP laptops. The ongoing concern of thermal runaway requires battery manufacturers to test all lithium ion batteries for weeks to ensure safety. Metallic impurities, not to be confused with metal oxide or salt impurities, in the part per billion level are known to cause an unacceptable number of batteries to experience thermal run-away in rechargeable Li-Ion batteries. The tolerances for these impurities are so low that there are no current analytical methods to measure. As such, all Li-Ion battery manufacturers test every battery manufactured for a few weeks to reduce, but not eliminate the occurrence of thermal run-away of production batteries prior to sale. This increases the overall cost of Li-Ion batteries, as the electronic equipment and labor involved is significantly higher than just simple materials production. What then is the solution to reducing thermal runaway, and improving lithium ion safety, and thereby cost efficiency? We believe the production of a cleaner, chemical manganese dioxide (CMD) and electrolytic manganese dioxide (EMD) is the answer.
Vancouver based American Manganese has provided Kemetco with manganese from their Artillery Peak project in Arizona. For this project, Kemetco has developed a patent pending hydrometallurgical process suitable for production of cleaner CMD and EMD materials. This hydrometallurgical process removes the crushing and grinding of the manganese ore body, which is the known source of impurities within finished manganese dioxide material. This hydrometallurgical process is what has allowed Kemetco to further our research into CMD and EMD material.
Hydrometallurgy Verses Grinding and Roasting

Hydrometallurgical processing for manganese production is certainly not a new concept. The idea has existed for decades, and yet the concept has struggled to reach commercially viable production. American Manganese and Kemetco research have taken the principal and adapted it with modern-day, commercially available processing equipment to create a unique hydrometallurgical process that is superior to current grinding and roasting processes.
The majority of current electrolytic manganese production comes from China where leaching of 10 percent to 12 percent carbonate ores requires a high quantity of sulfuric acid and South Africa, where conventional methods for refining manganese ore into electrolytic manganese involves high temperature roasting. Roasting operations can reach temperatures of up to 1,000°C, which is energy intensive. In order to remain economically feasible, this method works only for high-grade manganese deposits, generally more than 35 percent grades, and cannot support low-grade deposits.
With this new hydrometallurgical process, heat produced from the burning of sulphur to make SO2 provides additional energy for operating the hydrometallurgical process excluding electrolysis. The closed proprietary system of the hydrometallurgical process, along with the off-set of energy need with heat production means that the refinement of electrolytic manganese is low impact on the environment and requires lower energy costs.
Most importantly, the process removes the need for crushing and grinding, which eliminates the chance of metallic impurities entering into the processing stages of EMD and CMD material. It is through this advanced hydrometallurgical process that we are able to conduct research on advanced CMD and EMD material.
Researching Advanced Materials
There are several reasons why we believe our research will produce an improved battery material. First, since chemical manganese dioxide is precipitated as a powder directly from solutions, there will be no grinding of the product to powder form. Traditional grinding is known to introduce metallic impurities into the final product, which is known to cause internal short circuits in lithium ion batteries, leading to explosions and fires. Electrolytic manganese dioxide is electroplated as a thick hard sheet and must be ground to the right particle size to be put into a battery case.
Secondly, CMD can be produced much faster than EMD. Why? For one, there is no need for a grinding process that not only reduces the metallic impurities but also speeds up the processing time. CMD is precipitated instantly once an oxidant is added to the manganese containing solution whereas EMD requires electrowinning with a 24 to 48 hour harvest time.
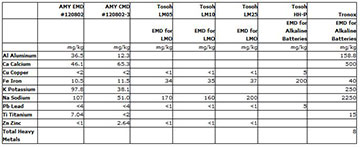
Our initial test results have shown positive improvements in the development of a clean CMD and EMD material. Samples of the initial EMD and CMD were analyzed by inductively coupled plasma spectroscopy (ICP) for cation impurities (not to be confused with metallic impurities), and compared to Tosoh and Tronox EMD products. Through this direct comparison we can see that our research has produced an EMD and CMD material with less cation impurities, specifically the lead and iron content (See Chart 1), while avoiding processes known to introduce metallic impurities Cation impurities can cause performance fade in lithium ion batteries, whereas metallic impurities can cause catastrophic failure resulting in explosion and fires.
These figures are the first set of results provided in the first quarter of our 12 month research program. We expect to see continued reduction in the metallic and mineral impurities as we move forward in our research.
The Case for CMD
Currently the market for Li-Ion batteries relies on a manganese dioxide spinel, which is supplied by the production of electrolytic manganese dioxide. EMD is the industry standard for lithium ion technology, yet it is far from perfect. EMD will always have metallic impurities due to the nature of processing; CMD however can eliminate steps known to introduce metallic impurities and significantly reduce the concern of thermal runaway.
The drawback with CMD is that while EMD is always electrochemically active, CMD is not always the case. It is important to properly process the final CMD product to ensure the material will function well in moving electrons. CMD that has been produced properly will ultimately hold a charge just as efficiently as EMD, be produced at a lower cost (CMD does not have to be put through a grinding process like EMD during the final stages of processing) and results in a cleaner product.
The cost savings of implementing clean CMD into Li-Ion batteries has significant potential. Take in consideration just the recalls mentioned earlier from this year. Nikon recalled 200,000 battery units for their digital SLR cameras, those cameras ranged in pricing anywhere from $1,200 to $3,000 a unit; Fisker Karma instituted a $55 million recall program for their electric vehicles supplied with A123 batteries. HP recalled 32,000 laptop batteries and in addition to the costs of recall and replacement, were also ordered to pay out a $425,000 settlement to claims regarding hazardous laptop incidents after the US Consumer Product Safety Commission charged the company for not issuing a recall quickly enough for knowingly selling laptops with batteries that could catch fire or overheat. While most of these past Li-Ion battery recalls (of various battery chemistries) were for consumer products, implementing practices to minimize battery failure is significantly more critical for mass use in electric vehicles due to the larger size of batteries packs and volume of production.
As the Li-Ion battery market continues to develop, and the demand for the product increases with the rise in electric vehicle adoption, the need for a cleaner and safer manganese material will be crucial. Kemetco’s research into CMD and EMD material is one step towards providing a marketable product that will advance Li-Ion battery technology and improve the overall cost efficiency and safety. We expect over the course of the next year to be able to provide research results that improve on current commercially available materials, and eventually lead to the cleanest form of CMD and EMD available on a global scale.
For more information, please visit www.kemetco.com.







